Proteus Digital Health fails to close a $100M funding round
The company aims to refocus on the most valuable near-term opportunities to deliver the promise of digital medicines for patients and their care teams.
FDA asks for more tests before it approves Proteus-Otsuka’s sensor-equipped pill
The two companies submitted the system as a new drug application (NDA) with the FDA in September 2015, and the Agency now declined to approve it.
Proteus Digital Health raises $50M Series H
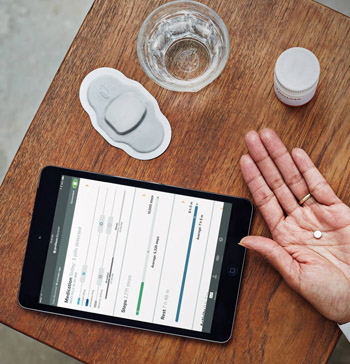
The company plans to drive increased adoption of its Digital Medicine offering, called Proteus Discover, by partnering with health system customers.
Chinese drug maker to invest $40M in Proteus Digital Health
As part of the deal, Gloria will also get an 18-month period of exclusive negotiation with Proteus to establish a joint venture company in China.
Barton Health first to implement Proteus’ digital health solution
Proteus' offering will be used to improve health outcomes for patients with long-term chronic conditions who are having challenges managing their medications.
First digital medicine starts its journey with the FDA
A drug/device product combines Otsuka's ABILIFY for serious mental illness, embedded with the Proteus ingestible sensor in a single tablet to record ingestion.
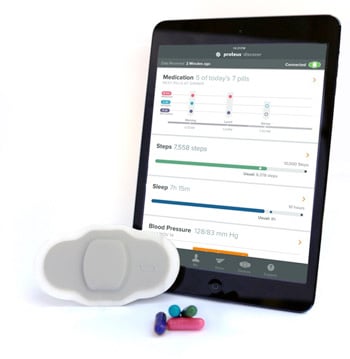
FDA says Proteus Ingestible Sensor can be used for medication adherence
When used with a medication, the Ingestible Sensor marks actual intake time, which is a quantifiable event that has allowed FDA to grant the expanded indication.
Oracle, Proteus join forces to help increase clinical trial accuracy
The platform is made to help health sciences organizations more effectively track and manage the use of medication during clinical trials.
Proteus Recovery is a biometrics monitoring patch for athletes
The device is touted as the "only system that supports the 24-hour optimization of athlete recovery and daily physiologic load."
Proteus Digital Health’s Series G round of funding reaches over $172 million
The new money will enable the company to advance the manufacturing and commercialization of its digital medicines.